Business
Heard on the Street Recap: Silver Scream
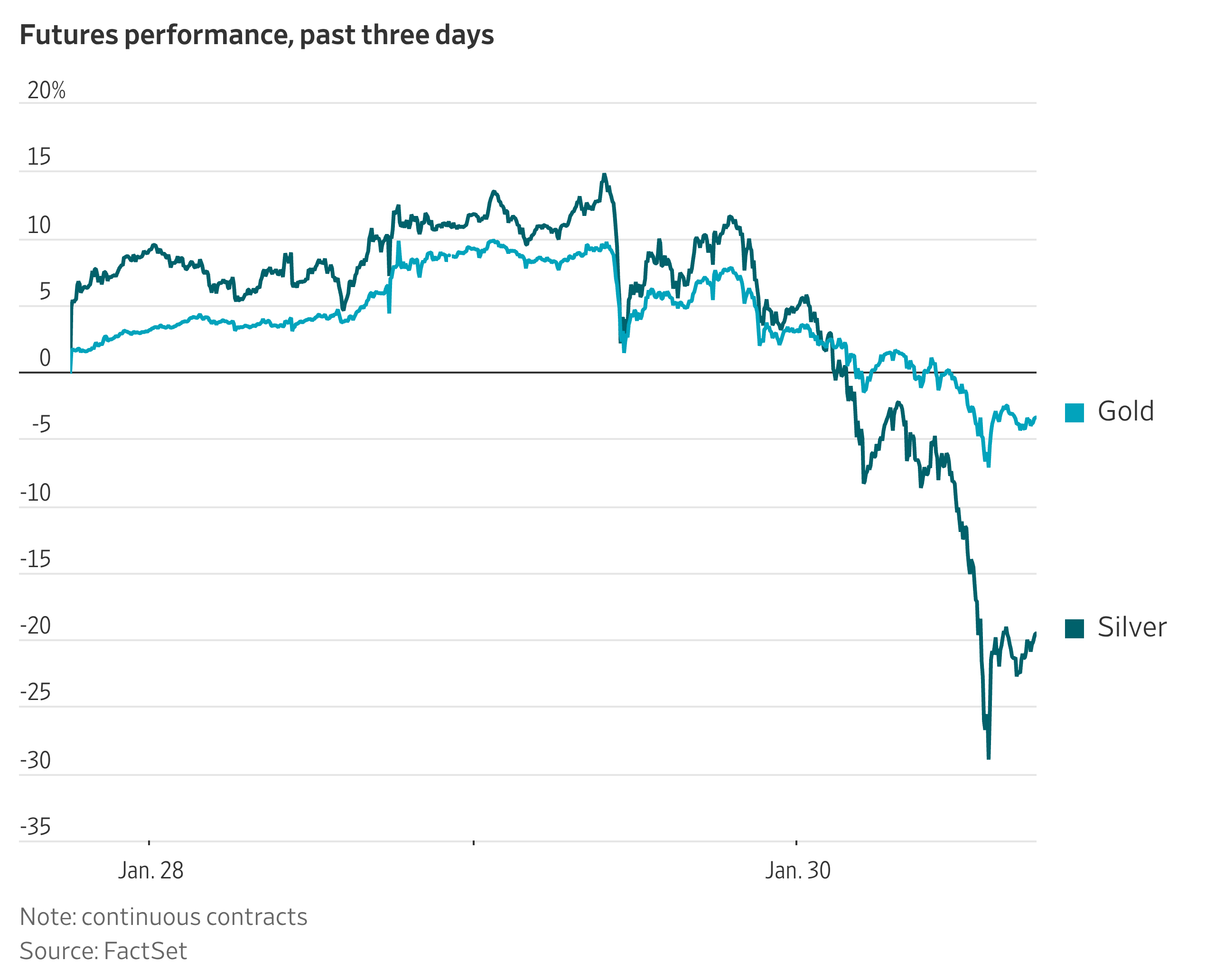
President Trump picked Kevin Warsh to be the next chairman of the Federal Reserve, choosing a former Fed official who has aligned himself with the president’s criticism of the central bank. Some market participants see Warsh as a relatively safe option, given his Fed experience and his track record as an inflation hawk. That could make him more resistant to calls from the administration to slash interest rates. President Trump said Warsh didn’t commit to cutting interest rates in an interview for the job, but he added “he certainly wants to cut rates.”
The announcement helped the dollar and hammered precious metals Friday. Silver prices crashed 31%, their second worst day on record. Gold plunged 11%. Both suffered their worst one-day slides since 1980. Silver has had a meteoric rise, smashing through a 45-year old record high in October and more than doubling from there. Earlier this month, Heard editor Aaron Back recommended selling silver.
Major U.S. indexes fell. The Nasdaq composite declined 0.9%, the S&P 500 and the Dow each fell 0.4%. Longer-term bond yields edged higher. Apple gained a meager 0.5% despite posting blowout iPhone sales and quarterly profit. Investors were concerned by rising costs, particularly for memory components.
Business
Eric Swalwell, candidate for California governor, denies sexual assault allegations


Eric Swalwell, candidate for California governor, denies sexual assault allegations
Business
Regulator bars 39 entities for alleged manipulation of RRP Semicon share price

The regulator said the stock has risen over 700 times in just 19 months. It said the company’s shares rose from ₹15 in April 2024 to ₹10,887 by October 2025, a surge that was not supported by its financials or business fundamentals.
During its investigation, Sebi observed that there was a coordinated network of promoters, preferential allottees, off-market transferees and a set of trading entities, who artificially inflated the share price of RRP Semiconductor.
The regulator said the scheme appears to have commenced with the acquisition of control of the company by Ira Mishra, the daughter of Ramesh Mishra, who was subsequently appointed as a director in the company. This was followed by a significant increase in the share capital of RRP through preferential allotment of shares to select entities.
The primary beneficiary of the allotment was one Rajendra Chodankar, who holds 74.5% of the share capital of RRP post allotment. Sumita Mishra, wife of Ramesh Mishra, was also a preferential allottee. Of the remaining preferential allottees, three were brought in by Ramesh Mishra and the rest byChodankar, it said.
The preferential allotment was accompanied by the company altering its MoA and changing its name to “RRP Semiconductor”, apparently to showcase its future forays into the promising semiconductor space. Simultaneously, the public shareholding (a small part of which was in demat mode) was fragmented and distributed through a chain of off-market transfers to multiple entities in small quantities, Sebi said in its order.
“The off-market transferees subsequently sold shares of RRP in minuscule quantities, largely to entities that consistently placed large buy orders at upper circuit limits and contributed significantly to the total market positive LTP. The trading pattern, characterised by insignificant sell quantities and aggressive buy orders at circuit limits, is prima facie not reflective of genuine market behaviour, but indicative of a pre-arranged and manipulative strategy to artificially inflate the price of the scrip of RRP,” Sebi whole-time member Amarjeet Singh said in his order.
Business
Is Taylor Swift Performing at Coachella 2026? Pop Icon Skips Desert Festival Amid Wedding Buzz and Tour Plans

Taylor Swift is not performing at the Coachella Valley Music and Arts Festival in 2026, despite persistent online rumors and fan speculation that the global superstar might make her long-awaited debut on the Empire Polo Club stages.

The 36-year-old singer-songwriter, one of the biggest names in music, has never performed at Coachella in her two-decade career — a fact that continues to fuel conjecture every spring. This year’s edition, running April 10-12 and April 17-19, features headliners Sabrina Carpenter on Fridays, Justin Bieber on Saturdays and Karol G on Sundays, with no slot or surprise appearance listed for Swift.
Fact-check reports and official lineup announcements have repeatedly debunked claims that Swift would step in, particularly after false rumors circulated suggesting Sabrina Carpenter might withdraw due to health issues or scheduling conflicts tied to Swift’s reported wedding plans with Travis Kelce. Those rumors proved entirely unfounded, with Carpenter delivering her highly anticipated headlining set as scheduled on Friday nights.
Swift’s absence aligns with her preference for large-scale, self-produced stadium tours rather than festival slots that often require scaled-back productions. Her Eras Tour, which wrapped in 2024 after breaking records worldwide, showcased elaborate staging, multiple acts and career-spanning sets that many industry observers say make festival appearances logistically challenging. Insiders note that Swift has consistently avoided one-off performances at events like the Super Bowl halftime show for similar reasons, prioritizing control over her artistic vision.
Recent speculation spiked in early April when unverified social media posts suggested Swift could replace Carpenter or appear as a special guest. Fact-checking outlets quickly clarified that Carpenter faced no reported health setbacks and remained committed to her Coachella headline slots. Swift has made no public statements about the festival, and Goldenvoice, the event’s promoter, has not included her in any official announcements or updates.
Swift and Kelce have a documented history with Coachella as attendees rather than performers. In 2024, the couple was spotted hand-in-hand enjoying sets by artists including Ice Spice and Bleachers, drawing massive media attention and cementing Swift’s status as a style and cultural influencer at the desert gathering. Pinterest data from recent weeks even showed Swift overtaking past festival fashion icons in search popularity, despite her non-performing role.
This year, as Weekend 1 unfolds, Swift remains focused elsewhere. Reports indicate she is deep into planning her next musical era, with speculation pointing to a potential new album in late 2026 or early 2027 followed by an international tour. Insiders close to the couple have also hinted at wedding preparations, with save-the-date cards reportedly circulating for a summer ceremony that could further limit her availability for festival appearances.
Coachella 2026 celebrates its 25th anniversary with a diverse, pop-heavy lineup that has drawn praise for its balance of emerging talent and established stars. Beyond the headliners, the bill includes The xx, Disclosure, Turnstile, Ethel Cain, The Strokes, David Byrne, Young Thug, FKA twigs, Wet Leg, Iggy Pop and a strong electronic contingent featuring Anyma, REZZ, Subtronics and Armin van Buuren. Late additions such as Jack White on the Mojave Stage have added further excitement.
Set times released earlier this week show music beginning around 1 p.m. daily, with headliners closing the Coachella Stage in prime evening slots. Extensive YouTube livestreams allow global audiences to tune in, a format that has proven popular even for non-attendees like Swift fans hoping for surprise cameos that never materialized.
Festival organizers emphasize safety, sustainability and immersive art installations alongside the music. Temperatures in the Coachella Valley remain a key consideration for attendees, with wide swings between daytime heat and cooler evenings. The official app and website provide real-time updates, stage maps and personalized schedules.
While Swift is not on stage, her cultural footprint looms large. Fans have speculated about possible guest appearances during Carpenter’s set — ranging from playful ideas like a Muppets crossover to more serious long-shot hopes of Swift joining for a duet. Carpenter herself has addressed the buzz lightly in pre-festival interviews, focusing instead on her own ambitious production, which she described as the most elaborate of her career.
The absence of Swift highlights broader conversations about festival economics and artist strategy. Coachella pays headliners handsomely but cannot match the revenue potential of a full Swift tour. Industry analysts point out that artists of Swift’s stature often generate more value through exclusive residencies, album cycles or stadium runs than through multi-weekend festival commitments that split focus and require production compromises.
Swift’s last major festival performance dates back years, with her career trajectory favoring self-contained spectacles. Her “Reputation” Stadium Tour, “Lover Fest” plans (later canceled due to the pandemic) and the record-shattering Eras Tour all underscored a preference for environments where she controls every element, from lighting to surprise songs to guest lists.
As Coachella Weekend 1 continues and Weekend 2 approaches, attention remains on the announced roster delivering memorable moments. Carpenter’s Friday sets have already sparked viral clips and social media praise for their energy and theatrical flair. Bieber’s Saturday debut and Karol G’s history-making Sunday close are expected to draw similar buzz.
For Swifties disappointed by the confirmation of her non-participation, consolation comes in the form of continued livestream access and the knowledge that new music may arrive soon. Rumors of a 2027 Coachella headline slot have circulated in fan communities, though nothing has been confirmed.
In the meantime, the desert festival proceeds without one of pop’s biggest names — a reminder that even in an event known for surprises, some absences are by design. Swift’s influence on music, fashion and fandom endures regardless, as evidenced by her continued dominance in cultural conversations surrounding the event.
Coachella 2026 runs through April 19. Tickets for Weekend 2 remain unavailable through official channels after selling out rapidly, though resale markets show fluctuating prices. Fans are advised to monitor official sources for any last-minute programming changes, though Swift’s involvement has been definitively ruled out.
The 2026 edition will be remembered for its strong pop representation and genre diversity, even as questions linger about when — or if — Taylor Swift will ever grace the Coachella stage.
Business
2nm A20 Chip, Bigger Battery and Variable Aperture Camera

CUPERTINO, Calif. — Apple’s iPhone 18 Pro Max is shaping up as one of the most significant upgrades in years, with rumors pointing to a powerful 2nm A20 Pro chip, a larger battery that could deliver record endurance, and a groundbreaking variable aperture camera system as the tech giant prepares for its fall 2026 lineup.

The high-end model is expected to launch alongside the iPhone 18 Pro and a new foldable iPhone in September 2026, while standard iPhone 18 variants may be delayed until spring 2027 in what could mark a major shift in Apple’s annual release cadence. With roughly five months until the anticipated unveiling, leaks from supply chain sources and analysts have painted an increasingly detailed picture of the device that could command premium pricing and push technological boundaries.
At the heart of the iPhone 18 Pro Max rumors is Apple’s leap to a 2-nanometer manufacturing process for the A20 Pro chipset. Built by TSMC, the advanced node is projected to deliver around 15% better CPU performance and up to 30% improved power efficiency compared with the 3nm A19 Pro in the current generation. The chip is also expected to feature enhanced memory bandwidth and stronger on-device AI capabilities, further supercharging Apple Intelligence features.
Battery life has emerged as a major talking point. Multiple leakers suggest the iPhone 18 Pro Max could house a 5,100 to 5,200 mAh cell — among the largest ever in an iPhone — potentially enabling up to 40 hours of mixed usage on a single charge. The increase is tied to a slightly thicker chassis, with reports indicating the device may measure about 8.8 mm thick and weigh over 240 grams, making it one of the heaviest iPhones in recent memory.
Design changes appear relatively modest overall. The iPhone 18 Pro Max is expected to retain the general aesthetic of the iPhone 17 Pro Max, including the same 6.9-inch LTPO OLED display with 120Hz ProMotion technology. However, the front could feature a slimmer Dynamic Island, with some Face ID components moving under the screen. Full under-display Face ID remains uncertain, with conflicting reports suggesting technical challenges may have delayed the complete removal of the cutout.
Camera upgrades could prove transformative. Rumors indicate the main rear camera may gain a variable aperture system — a first for iPhone — allowing users greater control over depth of field and light intake, similar to professional DSLR lenses. The triple-lens array is expected to stick with 48-megapixel sensors across wide, ultrawide and telephoto lenses, possibly incorporating a new three-layer stacked image sensor from Samsung. An 18-megapixel front camera is also in the mix.
Color options have generated excitement among fans. Bloomberg’s Mark Gurman reported that Apple is testing a “deep red” or burgundy finish for the Pro models, which would mark the first time a Pro variant comes in red. Other rumored hues include purple and brown, potentially replacing some of the iPhone 17 Pro’s palette. A semi-transparent or two-tone back glass design has also been mentioned, though major overhauls appear unlikely given the success of recent titanium builds.
Connectivity improvements are another focus. The iPhone 18 Pro Max could include Apple’s C2 modem with full satellite internet support via NR-NTN technology, enabling always-connected capabilities even without cellular coverage. Wi-Fi 7 and mmWave 5G are also expected, alongside possible refinements to the Camera Control button introduced in prior models.
Pricing remains a sensitive topic. The move to a costly 2nm process has raised concerns that the iPhone 18 Pro Max could become Apple’s most expensive iPhone yet, with some speculation of a noticeable increase over the current Pro Max starting price. Exact figures have not leaked, but analysts note the premium could reflect the advanced manufacturing and new features.
The broader 2026 iPhone strategy appears ambitious. With the standard iPhone 18 potentially pushed to 2027, Apple’s fall event could spotlight the Pro duo and the long-awaited foldable iPhone, creating a more premium-heavy lineup. This shift has fueled speculation about how the company will balance innovation with accessibility across its portfolio.
Industry observers say the rumored upgrades address common user requests. A bigger battery tackles one of the most frequent complaints about flagship phones, while the variable aperture camera could elevate mobile photography to new heights. Enhanced efficiency from the 2nm chip should translate to cooler operation and longer real-world usage, particularly for power-hungry tasks like video recording or AI processing.
Supply chain reports indicate early production planning is underway, though final specifications could still evolve. Apple has a history of refining features late in development, meaning some rumored elements — such as the exact extent of under-display Face ID integration — may not materialize as described.
For consumers weighing an upgrade, the iPhone 18 Pro Max rumors suggest meaningful improvements in performance, endurance and creative tools rather than revolutionary design changes. The device is likely to appeal strongly to professionals, content creators and power users seeking the best available iPhone experience.
As anticipation builds toward September, more concrete details are expected to surface through additional leaks and analyst reports. Apple has not commented on any upcoming products, maintaining its usual policy of secrecy until official announcements.
Whether the iPhone 18 Pro Max delivers on these expectations could influence not only Apple’s sales but also the broader smartphone market’s direction in 2026 and beyond. For now, the rumors paint an enticing picture of a refined flagship that prioritizes practical enhancements and cutting-edge internals.
Business
US, Iran set for peace talks but doubts emerge over Lebanon, sanctions


US, Iran set for peace talks but doubts emerge over Lebanon, sanctions
Business
McGraw-Hill: The EdTech Sleeping Giant

McGraw-Hill: The EdTech Sleeping Giant
Business
Stable Business Adapt During Times of Instability

Stable businesses come into their own during times of instability. Whatever causes market instability, well-equipped businesses find ways to adapt. This is particularly pertinent at the moment, given recent changes to UK tax policies.
The most recent example of Chancellor Rachel Reeves’ budgeting taking hold is the change to Remote Gaming Duty (RGD). The levy imposed on gambling operators in the UK jumped from 21% to 40% on April 1, 2026. According to the government, the increase is designed to reflect the growth of online casino gaming.
Reeves’ assessment is, indeed, correct. Gambling operators diversified their portfolios over two decades ago to align with changing tastes. For example, Paddy Power was formed in 1988 through the merging of three Irish bookmakers.
From a network of betting shops in Ireland, Paddy Power now has a global network of online and offline assets. The casino at Paddy Power alone gives players access to upwards of 2,000 games, including exclusives such as Paddy’s Mansion Heist. This asset sits alongside a sportsbook, poker site and bingo room, as well as live betting shops.
Nothing Changes by Staying the Same
Diversification was the strategy back then, and it will be once again as gambling operators respond to RGD increasing to 40%. Gambling operators aren’t the only ones facing tax hikes. The beleaguered hospitality sector is now contending with increased business rates and a rise in the minimum wage.
Talking to The Guardian, Nick Evans, co-owner of the Old Crown Coaching Inn in Faringdon, Oxfordshire, said he can’t increase prices any further. The former city trader is almost ready to admit defeat when it comes to the food and beverage side of his business.
“The only way you can make it work is to have a microwave and staff who can open a packet and put it on a plate. That’s not the reason we entered this industry”, Evans told The Guardian.
To keep his business afloat, Evans is doing what many old pubs are doing: he’s adding more hotel rooms. Adding six more rooms to the 14 already in place would “allow us to grow”, Evans explained. In this case, diversification is a necessity rather than a luxury. What’s important to take from this as a business owner is that standing firm isn’t necessarily the answer.
Cost-Cutting Isn’t Always the Answer
A recent report by the British Chambers of Commerce shows that 55% of UK businesses are increasing their prices as a result of tax hikes. A further 26% have cut their investment plans. Alongside those moves, finding new streams of income is crucial. Gambling and hospitality aren’t the only industries currently facing economic changes.
The long-term effects of US President Donald Trump’s war on Iran are already affecting haulage companies and farmers due to higher oil prices. These costs will filter through dozens of industries, meaning very few will escape.
Cost-cutting is a valid strategy, but so is diversification. Finding ways to add new services through online channels might be the way. Diversification could look at a new range of products. Whatever the pivot, it needs to address the central issue of raising costs by either increasing revenue or offering something at a lower price. The best businesses manage this, which is why they remain stable during unstable times.
Business
Guardrails for MFIs need to continue

The issue was discussed at a microfinance conclave organised by Sa-Dhan in Mumbai.
Microfinance guardrails in India, enforced by self-regulatory organisations such as the Microfinance Industry Network and Sa-Dhan, were implemented to ensure responsible lending and curb borrowers’ over-indebtedness, which was the primary reason for the severe stress the sector has been passing through over the past two years.
The key measures include limiting lenders to three per borrower, capping total indebtedness at ₹2 lakh, and restricting loans to customers with existing, significant delinquencies.
“The microfinance sector has shown resilience over the years, but it must continue to evolve with changing realities. By focusing on governance, transparency, and customer well-being, these guardrails will help build greater trust and long-term sustainability in the sector,” Sa-Dhan chairman K Paul Thomas said.
At the conclave, Reserve Bank of India’s central board member Satish Marathe is said to have suggested setting up a steering committee to address issues faced by the microfinance sector.
Business
Exclusive-Third Point won’t run proxy fight at CoStar, exits position, letter says


Exclusive-Third Point won’t run proxy fight at CoStar, exits position, letter says
Business
Trump Pressures Iran As Islamabad Talks Aim To Secure Lasting Middle East Truce

Trump Pressures Iran As Islamabad Talks Aim To Secure Lasting Middle East Truce
-

 Business5 days ago
Business5 days agoThree Gulf funds agree to back Paramount’s $81 billion takeover of Warner, WSJ reports
-

 Sports6 days ago
Sports6 days agoIndia men’s 4x400m and mixed 4x100m relay teams register big progress | Other Sports News
-

 Politics11 hours ago
Politics11 hours agoUS brings back mandatory military draft registration
-

 Fashion12 hours ago
Fashion12 hours agoWeekend Open Thread: Veronica Beard
-

 Business7 days ago
Business7 days agoExpert Picks for Every Need
-

 Tech3 days ago
Tech3 days agoHow Long Can You Drive With Expired Registration? What Florida Law Says
-

 Business6 days ago
Business6 days agoNo Jackpot Winner, Prize to Climb to $231 Million
-

 Fashion5 days ago
Fashion5 days agoMassimo Dutti Offers Inspiration for Your Summer Mood Board
-

 Sports12 hours ago
Sports12 hours agoMan United discover Nico Schlotterbeck transfer fee as defender reaches Dortmund agreement
-

 Fashion4 days ago
Fashion4 days agoLet’s Discuss: DEI in 2026
-

 Crypto World3 days ago
Crypto World3 days agoBitcoin recovers as US and Iran Agree a Ceasefire Deal
-

 Business9 hours ago
Business9 hours agoTesla Model Y Tops China Auto Sales in March 2026 With 39,827 Registrations, Beating Cheaper EVs and Gas Cars
-

 Crypto World2 days ago
Crypto World2 days agoCanary Capital Files SEC Registration for PEPE ETF
-

 Business6 days ago
Business6 days agoAkebia Therapeutics, Inc. (AKBA) Discusses Pipeline Progress and Strategic Focus on Kidney Disease Treatments at R&D Day – Slideshow
-

 Business17 hours ago
Business17 hours agoOpenAI Halts Stargate UK Data Centre Project Over Energy Costs and Copyright Row
-

 Tech5 days ago
Tech5 days agoHaier is betting big that your next TV purchase will be one of these
-

 Tech5 days ago
Tech5 days agoThe Xiaomi 17 Ultra has some impressive add-ons that make snapping photos really fun
-

 Tech5 days ago
Tech5 days agoSamsung just gave up on its own Messages app
-

 Tech5 days ago
Tech5 days agoGamer Restores the Original PlayStation Portal From Two Decades Ago
-

 Tech5 days ago
Tech5 days agoSave $130 on the Samsung Galaxy Watch 8 Classic: rotating bezel, sleep coaching, and running coach for $369











You must be logged in to post a comment Login