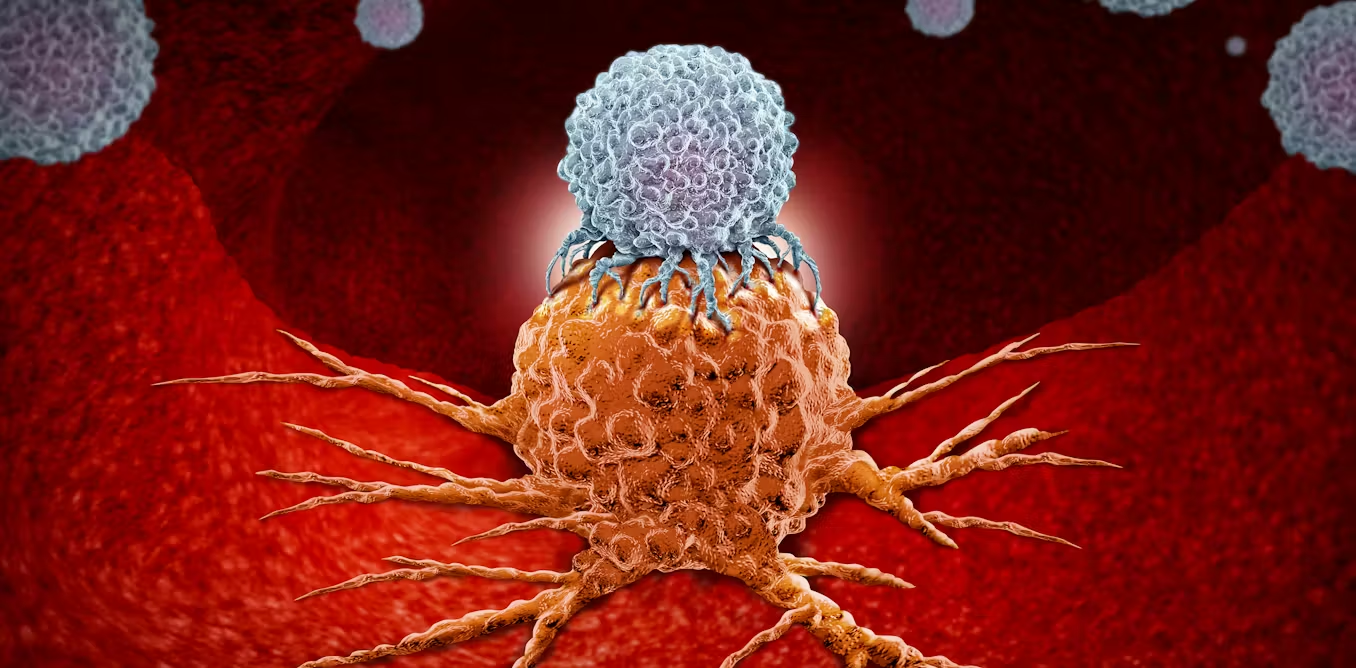
A new immunotherapy drug has demonstrated early promise in a recent prostate cancer clinical trial. The drug, called VIR-5500, is a “masked T-cell engager”. This type of immunotherapy ignites our own immune arsenal to fight cancer.
In the trial, which is still in progress and has not yet undergone peer-review, patients with advanced prostate cancer who had failed to respond to other treatments were given VIR-5500. Remarkably, initial findings showed that in the patients who received the highest doses, 82% saw reductions in their PSA (prostate specific antigen) levels – a commonly used measure of prostate cancer.
Strikingly, nearly half of the patients within this group also showed tumour shrinkage at both the primary tumour sites as well as in metastatic tumours (tumours which had spread from the prostate into different parts of the body).
Cancer cells have mechanisms to evade being eliminated by our immune system. But immunotherapies boost our immune system’s capacity to fight cancer. They do this by combatting these evasion strategies.
Various immunotherapies have demonstrated phenomenal success in recent years. Yet many cancers, such as prostate cancer, remain difficult to treat exposing the need for more effective immunotherapies.
T-cell engagers are a specific type of immunotherapy that works by anchoring immune cells, called T-cells, and cancer cells together by engaging molecules on the surface of both cell types. This enforced proximity prompts the T-cells to produce toxic cancer-killing chemicals and generate a cascade of inflammatory processes that promote cancer killing.
There are now over 200 different T-cell engagers, many of which are in clinical trials to treat a range of tumours including multiple myeloma, leukaemia and lung cancer.
T-cell engagers
T-cell engagers are not just being trialled for cancer. They may also help with treating other viral conditions, such as hepatitis B, which can cause life-long infection. As in cancer, the virus can evade our immune responses – but T-cell engagers can promote more effective clearance of virally-infected cells.
Despite the great promise surrounding T-cell engagers, the vigorous inflammation they trigger can also be a double-edged sword. In some cases, it can cause a serious inflammatory condition called cytokine release syndrome.
Cytokines are protein messengers released by cells that can drive inflammation. Normally, their release is tightly controlled – but in cytokine release syndrome, the response is excessive and uncontrolled. This can lead to multi-organ failure with potentially life-threatening consequences.
ALIOUI Mohammed Elamine7/ Shutterstock
Similar toxic inflammatory side-effects can be seen with other immunotherapies. It’s likely the condition is driven by the potent, acute activation of an immune response.
This is why T-cell engagers and other immunotherapy drugs need to be refined, to ensure their effects are less toxic.
One way of doing this involves producing versions of immunotherapies that are inactive but can be activated once inside tumours.
This is done by covering the drug in a “mask” that prevents it from engaging both the T-cells and cancer cells. When the drug enters tumours, molecules that are abundant in cancers can break down this mask, allowing the drug to engage its target cells. VIR-5500, the drug used in this recent, promising prostate cancer trial, is one of many new masked T-cell engagers.
As such, masking creates an effective drug that may also be safer. Tumour-specific activation should restrict the anti-cancer, inflammatory response to within the tumour, preventing widespread inflammation.
It may also enable the T-cell engagers to be more selective towards cancer cells, as some of their targets may also be expressed by normal healthy cells. This could simultaneously reduce toxicity and improve anti-cancer potency.
An additional benefit of masked immunotherapies is that the conversion from the inactive to active drug in the body takes time. This changes how the drug is dosed within patients.
In the clinic, T-cell engagers are often given in small doses that then need to be escalated to prevent acute immune over-activation. But the mask would allow the drug to be released more slowly, making delivery simpler and safer. The mask itself may also prevent the drugs from being broken down in the body and may extend their lifespan.
An important finding in this recent trial for prostate cancer was that most patients who received the highest doses of VIR-5500 suffered only mild inflammatory side effects. Given the known toxicity associated with T-cell engagers, this is an exciting finding – suggesting the masking is working to reduce the risks of excessive inflammation.
If further research proves that masking T-cell engagers creates safer, more effective drugs, then we can expand what we can do with them. They can be combined with more traditional cancer therapies, such as chemotherapy or radiotherapy, which may prove even more effective in eliminating cancer.
Other masked T-cell engagers have also shown early clinical promise in prostate cancer and trials have begun in numerous other cancers including pancreatic, colorectal and lung cancer.
As these trials are all ongoing, it’s too early to know the full extent of clinical success here. Early trials also only test within a small number of patients. The data has also not yet faced the scrutiny of peer-review and have only been presented at an oncology conferences.
Nevertheless, the initial results represent great hope for treating cancers that have proven otherwise difficult to treat with other immunotherapies.























![Trump's Worst FEAR Just Came True! [This Happens To Oil Next]](https://wordupnews.com/wp-content/uploads/2026/03/1773864099_maxresdefault-80x80.jpg)


































You must be logged in to post a comment Login