Business
Labor Department's proposal is a 'huge step' for your 401(k), BlackRock's Nefouse says

A proposed Department of Labor rule could significantly expand what Americans are able to hold inside their retirement accounts, potentially opening the door to assets like cryptocurrency, real estate and private markets.
BlackRock Global Head of Retirement Solutions Nick Nefouse described the rule as “a huge step forward for the 401(k) market” while discussing what the change could mean for everyday investors during his appearance on “Varney & Co.” Tuesday.
“The proposed regulation explains the steps that managers of 401(k) plans should take when considering alternative assets as a component in their investment lineups and establishes a set of process-based safe harbors for plan fiduciaries to use when selecting designated investment alternatives,” the Labor Department said in a press release on March 30.
Rather than endorsing specific investments, Nefouse suggested that the proposal is focused on creating a structured process for plan providers to follow when evaluating alternative assets.
AMERICANS TAP RETIREMENT FUNDS AT RECORD RATES AS MOUNTING FINANCIAL STRESS TAKES TOLL
“What the rule is trying to do… is establish a process, not necessarily say which asset classes are good or bad,” Nefouse said.
The shift could narrow a long-standing gap between retirement systems. While large institutional-style plans already have access to a wider range of investments, many workers in traditional 401(k) plans do not.
LARRY FINK CALLS FOR SOCIAL SECURITY REFORM, SAYS INVESTING A PORTION OF FUNDS COULD STRENGTHEN THE PROGRAM
“Think of regular people. About 25% of the population are in defined benefit plans. About 80% are in defined contribution plans,” Nefouse said.
“What we’re trying to do is level the playing fields, and so many Americans are relying on 401(k) plans,” he added.
The change could broaden access to investment options that have traditionally been limited to institutional retirement plans.
Business
Trump admin proposes opening 401(k)s to private equity, crypto


The Trump administration on Monday issued a proposed rule to allow retirement plans to offer alternative assets like private equity and cryptocurrencies as part of the investment options in 401(k) accounts.
The Labor Department’s rule aims to ease longstanding barriers to incorporating alternative assets into retirement plans and follows an executive order signed by President Donald Trump last summer on the subject.
Advocates for the rule change argue that including alternative assets in 401(k) plans can help foster better long-term returns and make diversification easier. Skeptics note that alternative assets can be less liquid, more complex and have higher fees, which can limit gains while also introducing risk.
Under the proposed rule, plan fiduciaries would have to objectively, thoroughly and analytically consider and make determinations about performance, fees, liquidity, valuation, performance benchmarks and complexity. Trustees who abide by those rules will be granted safe harbor that protects them from lawsuits.
TRUMP SIGNS ORDER TO OPEN 401(K)S TO PRIVATE MARKETS: WHAT IT MEANS FOR YOUR RETIREMENT
Managers of defined contribution plans have historically had the authority to consider alternative investments, though most have opted against doing so.
The Biden administration in 2022 issued a rescinded compliance release that warned fiduciaries against including cryptocurrency options in 401(k) plans, which the Trump administration criticized as a “departure from the department’s decades-long approach to fiduciary investment decisions.”
Labor Secretary Lori Chavez-DeRemer said that the agency’s newly proposed rule “will show how plans can consider products that better reflect the investment landscape as it exists today. This greater diversity will drive innovation and result in a major win for American workers, retirees, and their families.”
Treasury Secretary Scott Bessent added that the pending regulation “is an initial step in implementing the President’s Executive Order in a safe and smart manner, broadening access to additional retirement plan options for millions of Americans while being mindful of the importance of protecting retirement assets.”
NEW YORK INVESTMENT GIANT APOLLO JOINS HEADQUARTERS MIGRATION TO ‘FREEDOM’ STATES
Following the Labor Department’s release of the proposed rule, the agency will open a 60-day comment period ahead of a decision to finalize the rule.
Alternative asset managers like Blackstone and Apollo Global Management could benefit from the opportunity to draw on a new pool of capital. Several industry members and groups applauded the rule.
Apollo CEO Marc Rowan said that the change is a “thoughtful step toward addressing the growing retirement crisis,” noting that “Americans increasingly lack the savings and income needed for a secure retirement” and that the shift could “meaningfully improve retirement outcomes.”
GET FOX BUSINESS ON THE GO BY CLICKING HERE
If the rule is adopted, Erin Cho, a partner at the Mayer Brown law firm, said that it “will not open the floodgates for private equity, private credit or crypto funds to move into the retirement space” as it will only provide a process for doing so.
Reuters contributed to this report.
Business
Eli Lilly to acquire Centessa and sleep disorder drugs

Eli Lilly has agreed to pay up to $7.8 billion to acquire Centessa Pharmaceuticals and its experimental drug for excessive daytime sleepiness, the company said Tuesday.
Centessa is one of several companies working on a new class of drugs to treat narcolepsy, a condition that makes it difficult for people to stay awake during the day. The drugs may also be used to treat other neurological conditions that are accompanied by drowsiness, such as Alzheimer’s disease and depression, and possibly even more broadly.
Other possibilities include another severe sleep disorder called idiosyncratic hypersomnia, as well as other conditions where people experience sleepiness or executive function problems during the day and poor sleep at night, Lilly CEO Dave Ricks said in an interview with CNBC.
“We see a broader potential for this pathway, maybe a little bit of analogy to GLP-1, in a way that, you know, sleep and wakefulness are like core to our functioning, and when your sleep is disturbed or your wakefulness is disturbed, it causes a lot of other problems,” Ricks said. “So I think you can count on Lilly exploring broad use for [the orexins] and this new pathway, and we’re pretty excited about it.”
Under the terms of the deal, Lilly will pay $38 a share up front, or $6.3 billion for Centessa, a 38% premium to Monday’s closing price. If Centessa’s drugs win approval by the U.S. Food and Drug Administration by certain deadlines, Lilly will pay up to another $1.5 billion.
The transaction is expected to close in the third quarter, pending regulatory approval.
Shares of Lilly rose roughly 3% Tuesday, while Centessa’s stock surged 45%.
Orexin agonists used to treat narcolepsy and another severe sleep condition, called idiopathic hypersomnia, could amount to a $15 billion to $20 billion market if even about one-quarter of patients seek treatment, according to an estimate from Oppenheimer analyst Kostas Biliouris. Sales could go even higher if the drugs are used more broadly.
Centessa won’t be the first to market with its orexin agonist. A rival drug from Takeda is under review with the FDA and could be approved later this year.
Biliouris said he doesn’t expect Centessa’s drug to be approved until 2028, but he sees signs from mid-stage trial data that Centessa’s treatment could become the best in class.
Lilly, for its part, is a longtime leader in neuroscience. The company’s antidepressant Prozac catapulted Lilly to the top ranks of the pharmaceutical industry after it was approved in 1987.
More recently, Lilly introduced a drug called Kisunla for the early stages of Alzheimer’s disease with another trial on the horizon to see if the treatment can prevent the memory-robbing disease.
Lilly has been vocal about its intention to use the cash coming from its best-selling obesity and diabetes drugs Zepbound and Mounjaro to place more bets. Already this year, Lilly announced its intention to acquire cell-therapy company Orna Therapeutics and inflammation-focused Ventyx Biosciences.
Of the Centessa deal, Ricks said, “It’s the kind of thing we should be doing to really affect millions and millions of people, potentially, who suffer from neuroscience conditions like wakefulness and sleep.”
Business
WA, federal govt to sign enviro deal by year end, minister hopes

The state aims to enter a bilateral environmental regulatory agreement with the Commonwealth by late 2026, pushing out the timeframe previously flagged by Premier Roger Cook.
Business
Oil nears highest price since start of Iran war
The US-Israel Iran war has halted almost all traffic in a key waterway and the price Brent crude has surged.
Business
(VIDEO) Tiger Woods Had Hydrocodone Pills in Pocket During Florida DUI Rollover Crash, Affidavit Reveals

Golf legend Tiger Woods was found with two loose hydrocodone pills — a prescription opioid — in his left pants pocket after a single-vehicle rollover crash that led to his arrest on suspicion of driving under the influence last Friday, according to a probable cause affidavit released Tuesday by Martin County authorities.

The new details, first reported by TMZ Sports and confirmed across multiple outlets, paint a fuller picture of the March 27 incident on a residential road in the affluent Jupiter Island community. Woods, 50, told deputies he was distracted by his cellphone and changing the radio station moments before his luxury Land Rover struck the rear of a work truck and flipped onto its side. No one was seriously injured, but the crash has reignited questions about the 15-time major champion’s ongoing battles with pain management, prescription medications and road safety.
Deputies described Woods as profusely sweating despite cool air in the vehicle, moving in a “lethargic and slow” manner, and showing “severe signs of impairment.” When he removed his sunglasses, officers noted his eyes were “bloodshot and glassy” with “extremely dilated” pupils. A breathalyzer test registered 0.00 for alcohol, but Woods refused a urine test under Florida’s strengthened implied consent law for suspected drug impairment, leading to an additional charge.
During a search incident to arrest, deputies discovered two white pills marked “M367” in Woods’ pocket. The imprint identified them as hydrocodone, an opioid commonly prescribed for severe or chronic pain. Woods acknowledged taking “a few” prescription medications earlier that morning when asked by investigators. The pills were seized and entered into evidence.
The affidavit adds to the narrative of a golfer whose body has endured years of punishing physical tolls from elite competition and multiple surgeries, including multiple back operations and a 2021 car crash in California that left him with severe leg injuries. Woods has been open in the past about relying on pain medication during recovery periods, but the presence of loose opioids during a driving incident has drawn sharp scrutiny.
Martin County Sheriff John Budensiek said at a news conference last week that investigators believed Woods was impaired by “some type of medication or drug” rather than alcohol. Woods was booked on misdemeanor charges of DUI with property damage and refusal to submit to a lawful test. He was released from jail overnight Friday after posting bond and has not yet entered a plea.
The crash occurred around 2 p.m. as Woods reportedly attempted to pass a pressure cleaner truck on a road with a 30 mph speed limit. He crawled out of the overturned vehicle through a window and was seen on his phone near the wreckage. Photos from the scene showed the Land Rover resting on its side with visible damage.
This marks Woods’ second high-profile DUI-related incident. In 2017, he was arrested in Florida after being found asleep at the wheel of his Mercedes. Toxicology reports at the time revealed five substances in his system, including the opioid hydrocodone (Vicodin), hydromorphone (Dilaudid), Xanax, Ambien and THC. Woods later pleaded guilty to reckless driving and completed a program addressing his issues with prescription medications.
Tuesday’s affidavit has prompted fresh discussion about prescription opioid use among athletes managing chronic pain. Hydrocodone is a Schedule II controlled substance with high potential for dependence. Medical experts note that while it can be legitimately prescribed, combining it with other medications or using it while driving can significantly impair judgment, reaction time and coordination.
Woods has been attempting a cautious comeback in 2026 after years of limited competitive play due to injuries. He participated in the TGL Finals earlier in the week, a tech-infused golf league event, and had expressed hope of competing at the Masters Tournament, which begins April 9 in Augusta, Georgia. His representatives have not commented publicly on the latest developments or his plans for the storied event, where he has won five green jackets.
PGA Tour officials and fellow players have offered measured responses, emphasizing support for Woods’ health while noting the seriousness of impaired driving. Some teammates in the TGL expressed concern over what one called “disturbing” recent events.
The incident also revives memories of Woods’ 2021 rollover crash in Rancho Palos Verdes, California, where his SUV veered off a winding road at high speed. He suffered compound fractures in his right leg and underwent extensive rehabilitation. An empty pill bottle was reportedly found in that vehicle, though no charges were filed related to impairment.
Legal analysts say the current case could hinge on field sobriety test performance, the deputy’s observations of impairment, the refused urine test and any eventual toxicology results if pursued. Under Florida law, refusal to submit to testing after a DUI arrest can lead to automatic license suspension and may be used as evidence in court.
Woods owns multiple properties in the Jupiter area, including a waterfront estate, and has deep ties to South Florida’s golf community. He has largely kept a low profile off the course in recent years, focusing on his children, business ventures such as his golf course design firm and the TGL league he co-founded with Rory McIlroy.
Public reaction on social media has been swift and divided. Some fans expressed disappointment and concern for Woods’ well-being, while others highlighted the potential dangers of driving while impaired by any substance. The story dominated sports headlines Tuesday, with the new affidavit details amplifying coverage of the Friday crash.
As of Tuesday afternoon, no court date had been set. Woods remains eligible to travel and compete pending resolution of the charges, though any conviction could carry consequences for his driving privileges and public image.
The golf world will watch closely how Woods navigates this latest chapter. His resilience through physical adversity has been well-documented, but repeated incidents involving driving and medications raise questions about long-term management of his health and lifestyle.
Martin County authorities have not released bodycam or dashcam footage, citing the ongoing investigation. The second driver involved in the crash was not injured and cooperated with investigators.
For a figure who transcended golf to become one of the most recognizable athletes globally, Tuesday’s revelations add another complex layer to a career marked by triumph, scandal, injury and remarkable comebacks. Whether this proves a minor legal hurdle or a more significant turning point remains to be seen as the legal process unfolds.
Woods’ team has historically emphasized privacy around medical matters. In past statements, he has credited surgery, physical therapy and mental focus for his recoveries while acknowledging the cumulative wear on his body from decades at the highest level of professional golf.
As spring arrives and the golf season intensifies toward the Masters, the focus for many will shift from Woods’ past glories to his present challenges — both on and off the course. Supporters hope for transparency, accountability and continued progress in addressing any underlying issues with pain and medication.
Business
At Close of Business podcast March 31 2026

Isabel Vieira and Mark Pownall discuss a restructure with WA’s biggest wine maker Fogarty Wine Group.
Business
Apartments planned above resort’s iconic former Woolworths store


Amendments made to project at Art Deco landmark
Business
Perth family law firm Paterson & Dowding wins injunction to protect stolen data

A boutique Perth family law firm has been granted an injunction over data stolen during a recent cyber attack, preventing it from being accessed and shared by Australian outlets or companies.
Business
US petrol price tops $4 for first time since 2022

The Iran war continues to push up prices at the pump for US motorists.
Business
Novo Nordisk launches Wegovy subscription for GLP-1 obesity drugs
Wegovy semaglutide tablets.
Michael Siluk | Universal Images Group | Getty Images
Novo Nordisk on Tuesday launched a multi-month subscription program for its Wegovy obesity drug products that aims to ensure cash-paying patients see lower, “predictable” monthly prices.
Eligible patients can choose between three-, six- or 12-month subscriptions for the Wegovy injection or the two highest doses of the newly launched pill under the same brand name. Longer plans offer lower monthly pricing, and the company expects people to save up to $1,200 a year on the injection and as much as $600 a year on the pill, relative to paying for their individual dose each month, according to a Novo release.
Patients can expect to pay flat monthly prices, even if they move to different doses, the company said. The subscription program will be available starting Tuesday on several of Novo’s telehealth partners, including Ro, WeightWatchers, LifeMD, Sesame and Hims & Hers, with more expected to be added soon.
The first-of-its-kind offering is “an opportunity to help patients not only start but stay on therapy and help them manage the ups and downs of some of the pricing considerations,” regardless if they are starting treatment or are currently taking the drug, said Ed Cinca, Novo’s head of marketing and patient solutions.
Inability to stay on GLP-1s is a longstanding issue due to factors such as difficulty accessing the drugs and gastrointestinal side effects, with one 2025 study estimating that around 65% of patients with obesity stop treatment within a year.
Wegovy subscription prices and estimated savings
Injection subscription plans (0.25, 0.5, 1.7 and 2.4 milligram doses)
- 3-month: $329 per month, savings of $240 per year
- 6-month: $299 per month, savings of $600 per year
- 12-month: $249 per month, savings of $1,200 per year
Pill subscription plans (9 and 25 milligram doses)
- 3-month: $289 per month, savings of $120 per year
- 6-month: $269 per month, savings of $360 per year
- 12-month: $249 per month, savings of $600 per year
The new program also comes as Novo’s pill, which has seen explosive uptake since its U.S. launch in January, is set to face fresh competition from an upcoming oral GLP-1 from chief rival Eli Lilly later this year. Lilly is currently the dominant player in the branded GLP-1 market in the U.S., with an estimated 60% share, while Novo has about 39%.
The Wegovy pill has largely been reaching people who didn’t previously take GLP-1 injections, making it crucial for Novo to capture as many new patients as it can before a competitor arrives.
As Novo Nordisk’s subscription plans launch, cash-paying patients can still pay $149 per month for the lower doses of the pill, which are 1.5 and 4 milligrams. But starting in August, the 4-milligram dose will cost $199 per month. Meanwhile, the recently approved 7.2-milligram dose of Wegovy will be added to the subscription program at a later date.
Cinca emphasized that patients can opt out of the subscription while it’s active if they no longer wish to enroll.
“We want to help patients identify a path that can help them feel comfortable about treating [obesity] in the long term,” he added.
Cinca said Novo is not yet offering the program on its NovoCare direct-to-consumer pharmacy, but added that there’s “an opportunity to evaluate how this goes and then build it out” through that platform over time.
-

 NewsBeat6 days ago
NewsBeat6 days agoManchester United reach agreement with Casemiro over contract clause amid transfer speculation
-

 News Videos6 days ago
News Videos6 days agoParliament publishes latest register of MPs’ financial interests
-

 NewsBeat4 days ago
NewsBeat4 days agoThe Story hosts event on Durham’s historic registers
-
 Tech5 days ago
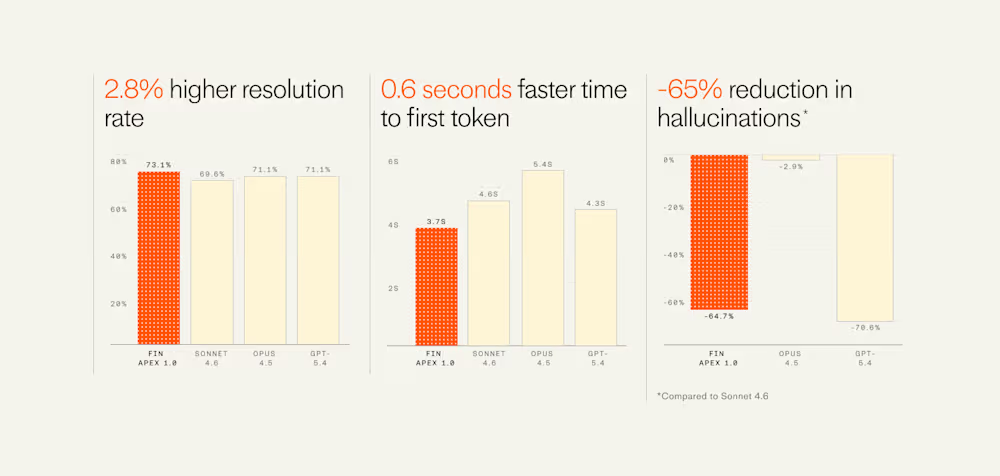
Tech5 days agoIntercom’s new post-trained Fin Apex 1.0 beats GPT-5.4 and Claude Sonnet 4.6 at customer service resolutions
-

 Sports4 days ago
Sports4 days agoSweet Sixteen Game Thread: Tide vs Michigan
-

 Business5 days ago
Business5 days agoInstagram, YouTube Found Responsible for Teen’s Mental Health Struggle in Historic Ruling
-

 NewsBeat6 days ago
NewsBeat6 days agoTesco is selling new Cadbury Dairy Milk bar and people can’t wait to try it
-

 NewsBeat7 days ago
NewsBeat7 days agoEntrepreneurs Forum survey reveals optimism in North East
-

 Entertainment3 days ago
Entertainment3 days agoLana Del Rey Celebrates Her Husband’s 51st Birthday In New Post
-
Entertainment1 day ago
Fans slam 'heartbreaking' Barbie Dream Fest convention debacle with 'cardboard cutout' experience
-
Crypto World8 hours ago
Dems press CFTC, ethics board on prediction-market insider trades
-

 Sports7 days ago
Sports7 days agoFantasy Baseball Week 1 Preview: Top sleeper hitters for both five- and 12-day period led by Munetaka Murakami
-

 Tech7 days ago
Tech7 days agoUS FCC Prohibits Approval Of New Foreign-Made Consumer Routers
-

 Sports3 hours ago
Sports3 hours agoTallest college basketball player ever, standing at 7-foot-9, entering transfer portal
-

 Tech7 days ago
Tech7 days agoEmbedding compliance in AI adoption
-
Sports7 days ago
One Team in Particular Is Monitoring Kirk Cousins
-

 Fashion7 days ago
Fashion7 days agoAlthea Mink: Patrice Taylor’s Atlanta Couture Brand Built on Legacy, Resilience, and Architectural Glamour
-

 NewsBeat7 days ago
NewsBeat7 days agoDWP to close two benefits for all new and existing claimants before April
-

 Fashion6 days ago
Fashion6 days agoHow to Style Spring Like WeWoreWhat: Easy Outfit Ideas for 2026
-
 Entertainment6 days ago
Entertainment6 days agoHBO’s Harry Potter Series Will Definitely Fail For One Big Reason, And It’s Not J.K. Rowling Or Snape










You must be logged in to post a comment Login