‘Our shared hope is that much of the valuable growth, innovation and inclusion work undertaken by Engine Shed over the years will continue’
Bristol work, meeting and event space Engine Shed is being closed after 13 years. The innovation hub next to Temple Meads station is to shut its doors for good in December, it has confirmed in an announcement on LinkedIn.
Engine Shed is run by a subsidiary which is wholly owned by the University of Bristol and the closure reflects “a shift in the university’s innovation activity”, according to the post.
The historic building, which was designed by Isambard Kingdom Brunel, is used by Engine Shed members for working, events and training through a membership scheme. It is also home to tech incubator SETsquared Bristol.
“There is no easy way to say it, after 13 years of catalysing Bristol’s innovation ecosystem, Engine Shed will close in December 2026,” the statement read.
“Many consider Engine Shed to have been both the spark that ignited much of the current innovation ecosystem and the beating heart that keeps collaboration, innovation and generosity central to our collective work.”
It is understood the team behind Engine Shed will move out of the property and will help run part of the main academic building on the new Temple Quarter Enterprise Campus. Staff who are part of SETsquared Bristol will relocate too.
“Our shared hope is that much of the valuable growth, innovation and inclusion work undertaken by Engine Shed over the years will continue through activities in the main academic building on the Temple Quarter Enterprise Campus (TQEC),” the LinkedIn post added.
Engine Shed is currently part of Bristol Innovations – the University of Bristol’s business support arm – and sits at the heart of the city’s Bristol Temple Quarter redevelopment, an ambitious urban regeneration project. Its partners include techSPARK, Barclays Eagle Labs, SETsquared Bristol and the Quantum Technologies Innovation Centre.
Engine Shed founder Nick Sturge said: “The ecosystem, and therefore the number and strength of the actors within it, has grown significantly since 2013 and I’m super proud that Engine Shed, and the team behind and within it, has played a part in that growth and the collaborative spirit that has created the right environment for the next stage of Engine Shed’s journey.
“While part of that will rest in the sidings for a while, I am so pleased that Bristol Innovations Zone, in the new TQEC building, will pick up the momentum that Engine Shed helped create.”
Emily Kent, co-founder of Bristol software firm One Big Circle, wrote in response to the LinkedIn post: “Brilliant team at Engine Shed over the years and an inspiring building. From our first meeting with our SETsquared Bristol mentor in 2014, our first designated desk, then four moves between ever larger office spaces, alongside so many networking events, panels, opportunities to present to fellow entrepreneurs, ministers and a fair few school visits to inspire the nextgen, Engine Shed has been a huge part of our OBC journey.
“We’re still near neighbours as we’ve grown and are very sorry to hear the doors will be closing but hope the spirit, ethos and culture of [Engine Shed] will travel well to its new home.”































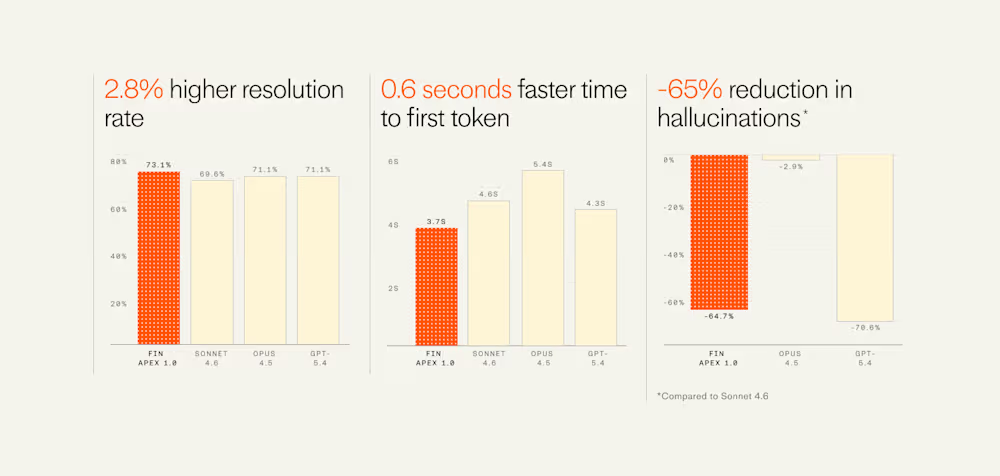
























You must be logged in to post a comment Login